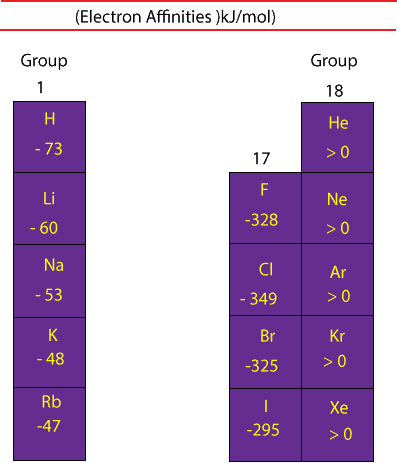
Chlorine most strongly attracts extra electrons. Nonmetals like to gain electrons to form anions to have a fully stable electron shell. Nonmetals: Generally, nonmetals have more positive electron affinity than metals.Mercury most weakly attracts an extra electron. The electron affinity of metals is lower than that of nonmetals. The second (reverse) definition is that electron affinity is the energy required to remove an electron from a singly charged gaseous negative ion. First, as the energy that is released by adding an electron to an isolated gaseous atom. Metals: Metals like to lose valence electrons to form cations to have a fully stable shell. Electron affinity can be defined in two equivalent ways.Chlorine most strongly attracts extra electrons, while neon most weakly attracts an extra electron.Īffinities of Non metals vs. Atoms whose anions are more stable than neutral atoms have a greater affinity. Moreover, nonmetals have more positive affinity than metals. However, nitrogen, oxygen, and fluorine do not follow this trend. The electron affinities will become less negative as you go from the top to the bottom of the periodic table. Generally, the elements on the right side of the periodic table will have large negative electron affinity. Although affinity varies greatly across the periodic table, some patterns emerge. The greater the negative value, the more stable the anion is. This affinity is known as the second electron affinity and these energies are positive.Įlectron affinity can be either positive or negative value. However, more energy is required to add an electron to a negative ion which overwhelms any the release of energy from the electron attachment process. By convention, the negative sign shows a release of energy. This affinity is known as the first electron affinity and these energies are negative. When an electron is added to a neutral atom, energy is released.

To use electron affinities properly, it is essential to keep track of sign. S + e – → S – – ∆H = Affinity = 200 kJ/mol Electron affinities are more difficult to measure than ionization energies.Īn atom of Sulfur in the gas phase, for example, gives off energy when it gains an electron to form an ion of Sulfur. Ionization energy is defined as the energy needed to remove one electron. In other words, the neutral atom’s likelihood of gaining an electron. Note that, ionization energies measure the tendency of a neutral atom to resist the loss of electrons. Electron affinity is defined as the change in energy (in kJ/mole) of a neutral atom (in the gaseous phase) when an electron is added to the atom to form a negative ion. In other words, it can be expressed as the neutral atom’s likelihood of gaining an electron. The change in energy (in kJ/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion.

In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Electron affinity of Sulfur is 200 kJ/mol.
